

Keelvar for pharmaceutical companies












What other pharmaceutical companies get from Keelvar
savings across $1bn spend
of events automated
average savings
events across 6 categories
Better sourcing outcomes for pharmaceuticals

Make award decisions based on total value, not just price
Model scenarios across cost, quality, compliance, supplier risk, and continuity — so every award reflects what the business actually needs, not just what came in lowest.
API availability and supplier capacity change fast. Trigger new sourcing events automatically — without waiting for a shortage to hit your formulary.
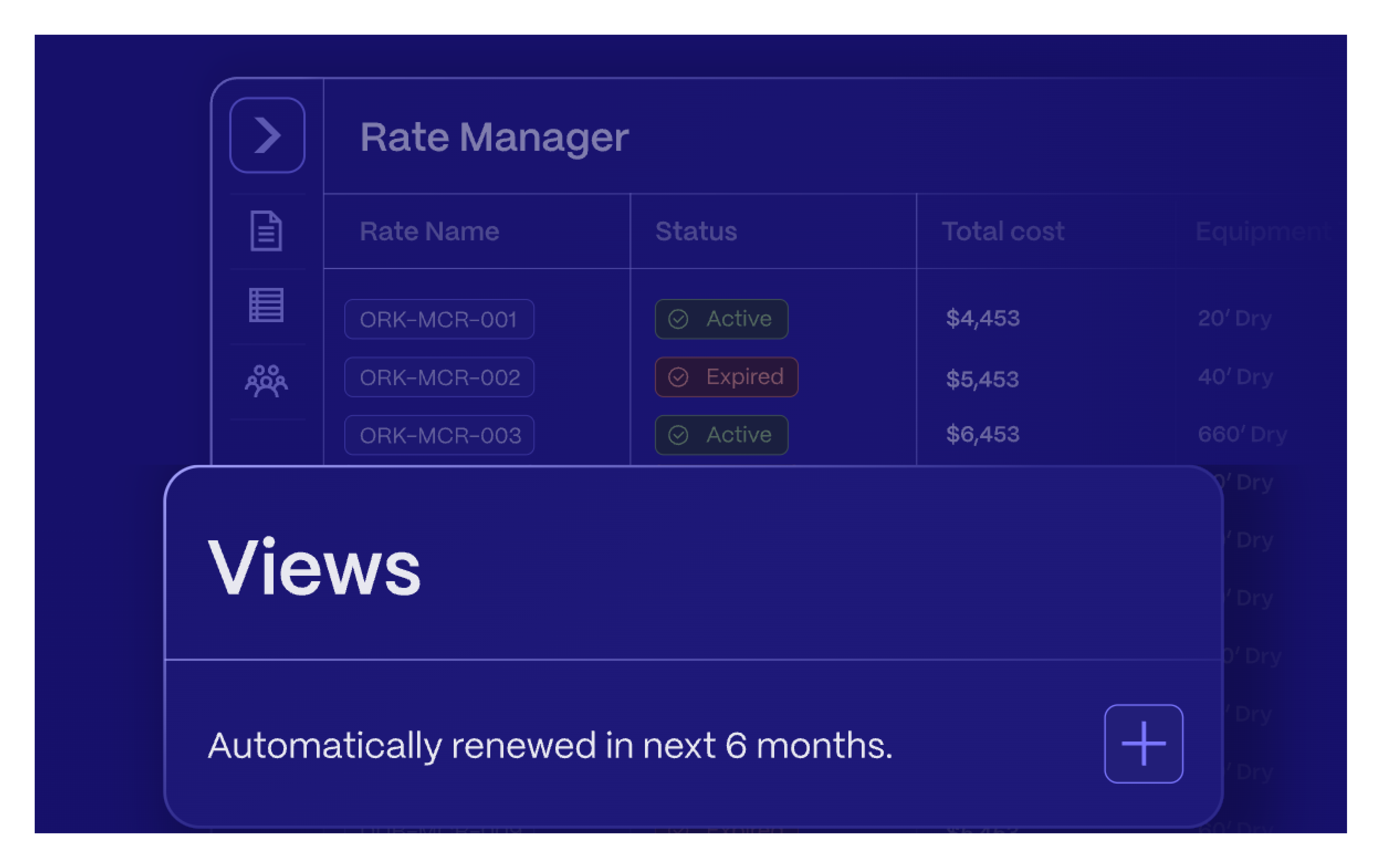
Build best practices once, apply them everywhere
Create workflows for key categories — APIs, excipients, packaging, contract manufacturing — and reuse them across sites, markets, and every sourcing cycle.

Run more events without adding headcount
Automate bid invites, reminders, validation, and conditional awarding. Your team focuses on decisions — not administration.


Suppliers see where they stand in real time — driving better bids across every event you run.
Do you need a product that can help you with sourcing?
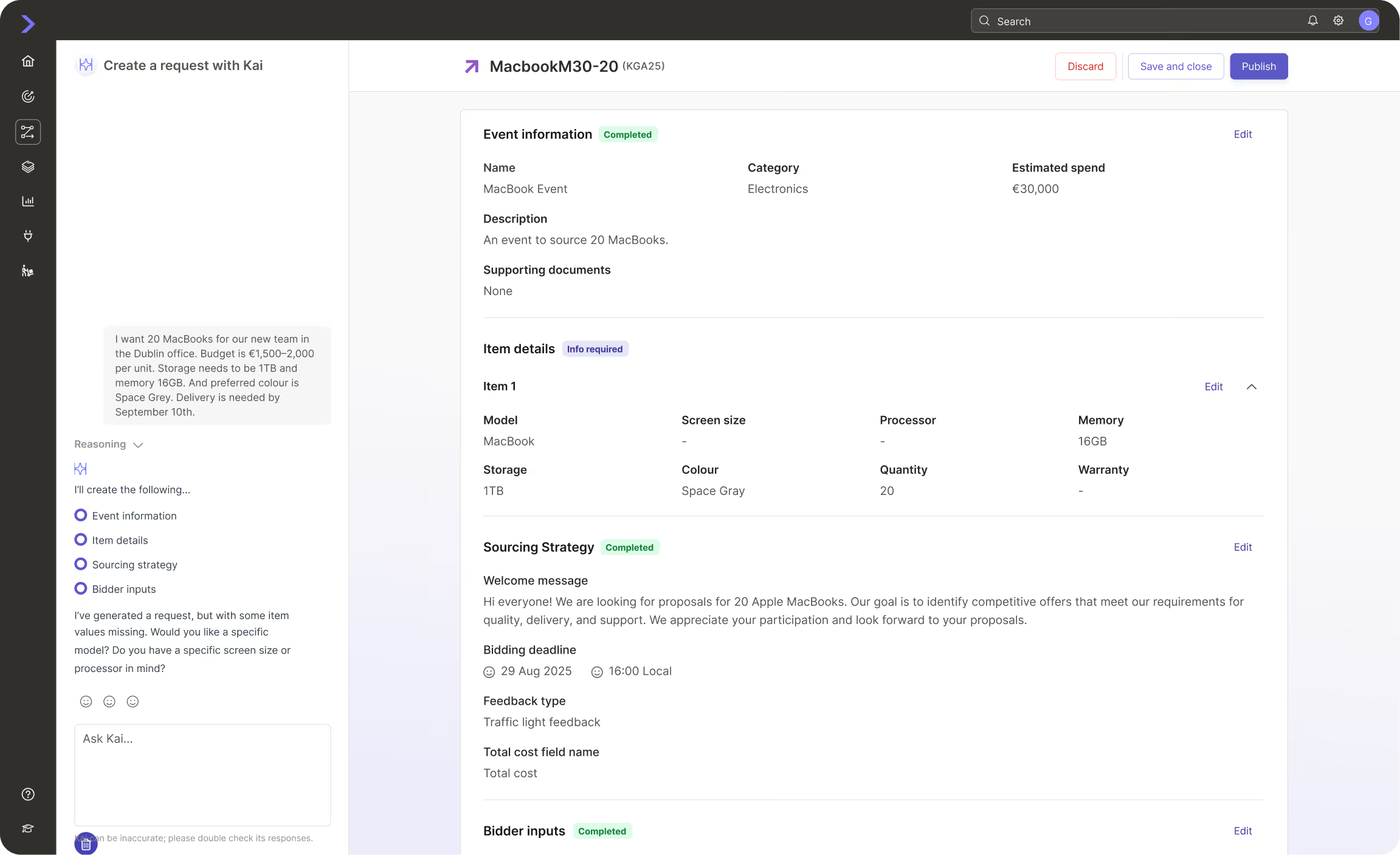
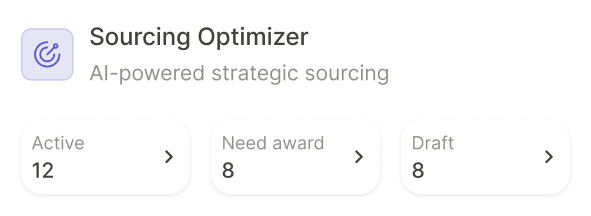

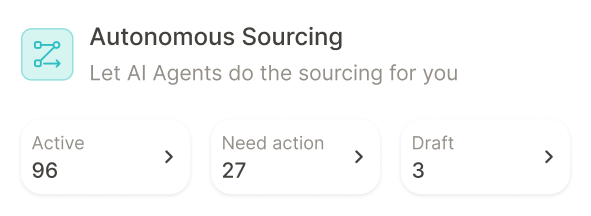

Where pharmaceutical companies are using Keelvar
See how other companies in your industry are using Keelvar across multiple spend categories.
Read moreWhat they say about us

Get in touch

FAQs
Straight answers to common questions about Keelvar.
Pharmaceutical procurement operates under strict compliance requirements, with complex direct materials sourcing, high-value indirect categories, and sophisticated supplier qualification processes. Keelvar is used in pharmaceuticals to run structured RfP and RfQ events with full audit trails, model complex award scenarios for API and packaging categories, and automate high-volume indirect sourcing while maintaining governance standards. A healthcare technology company using Keelvar cut time-to-bid by 90%.
Every sourcing event in Keelvar generates a complete audit trail: supplier invitations, bid submissions, bid revisions, scenario analysis, and award decisions. Autonomous sourcing workflows operate within rules defined by your compliance team, and human approval steps can be embedded at any stage. This means pharmaceutical procurement teams can move faster without sacrificing the documentation and process control regulators require.
Yes. Direct materials in pharmaceuticals — APIs, excipients, primary packaging — involves highly constrained supplier bases, regulatory requirements, and multi-site supply considerations. Keelvar's Sourcing Optimizer allows teams to model award scenarios that factor in supplier qualification status, geographic redundancy, and volume commitments alongside price. This means teams can identify the optimal commercial outcome while satisfying supply security and compliance requirements.
Indirect categories in pharmaceuticals — lab consumables, facilities services, logistics — are often high-volume and managed by lean teams. Keelvar enables these categories to be handled through configured, repeatable workflows that run with minimal manual input. Category managers set the rules; AI agents execute the events. This creates sourcing coverage that would not be commercially viable to achieve with manual resource alone.
.svg)






